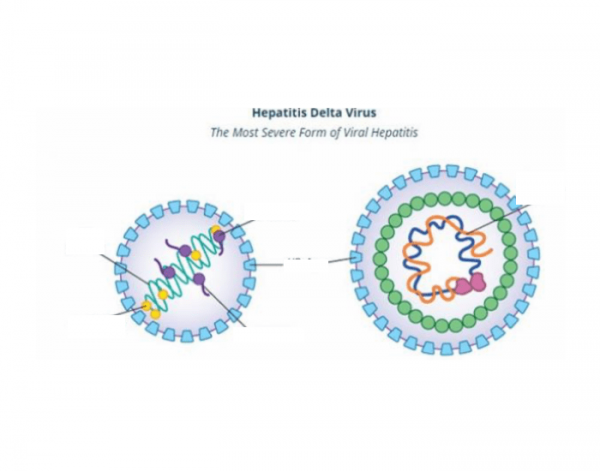
BioPharmaceuticals, Inc. (Nasdaq: EIGR), focused on the development and exploitation of targeted, first-in-class therapies for serious rare and ultra-rare diseases, these days proclaimed that a late-breaker speech has been granted for interim finish of treatment (Week 24) results of section 2 elevate (Peginterferon Lambda or Lambda) study at AASLD 2019 in state capital. Lambda could be a first-in-class sort III antiviral agent in clinical deve....
Tags : antiviral agent , Peginterferon Lambda, Combination Therapy, Chronic Hepatitis Delta Virus, Hepatitis Delta Virus,

comments (0)