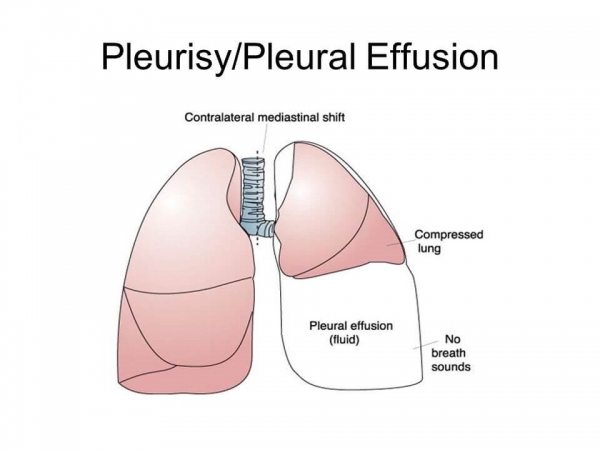
According to an announcement by an international biotechnology company in China, Clover Biopharmaceuticals, thriving to develop new biological therapies, the first-ever cancer patient suffering from malignant pleural effusions was admitted in Australia in a fully human TRAIL-Trimer fusion protein, the phase I of SCB-313 trial. Five clinical studies to evaluate SCB-13 and recruit patients from Australia and China.
Director of Pleural ....
Tags : Phase 1 SCB-313, Malignant Pleural Effusion , Treatment , Clover Biopharmaceuticals ,

comments (0)