Highlights
- The FDA has issued an emergency use authorization for it
- President Trump considers it a breakthrough
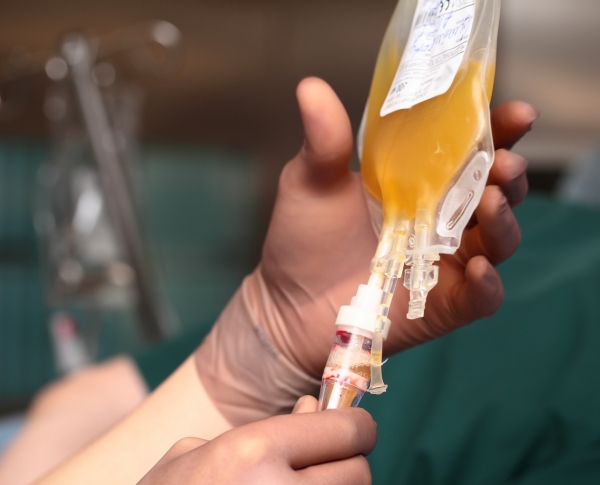
The Food and Drug Administration issued an emergency use authorization for the convalescent plasma treatment. It allowed US health-care providers to treat suspected or laboratory-confirmed Covid-19 in hospitalized patients.
&....
Tags : Food and Drug Administration, convalescent plasma treatment, President Donald Trump, World Health Organization, Corona virus patients ,

comments (0)